ChiroBlock's SupplyChainFactory
ChiroBlock's SupplyChainFactory Contract manufacturing of exclusive fine chemicals for your supply chain
Within our SupplyChainFactory, we manufacture exclusive fine chemicals on a regular basis for our customers, providing the missing link between the R&D results of our other activities and the manufacturing industry.
This chemical sourcing service bridges the gap between the lab-scale and multi-ton (per year) contract manufacturing.
Combining our large experience in chemical synthesis R&D with an integrated quality management system, we are your reliable source of complex and crucial compounds for your supply chain.
Strict control of starting materials, usage of dedicated equipment, implemented line clearance procedures, communication rules for change management and absolute traceability are essential features for customers seeking a reliable source for continuous supply, which is particularly important for compounds that are customer exclusive and not commercially available elsewhere.
Together with our customers, we define the requirements to find the perfect fit for your sourcing needs. From the beginning to the end, communication and mutual trust form the basis of consistent quality.
Chemical Sourcing - Manufacturing of exclusive fine chemicals
At first sight, the provision of chemicals to a customer's supply chain is not a new idea and is not worth paying much attention to, being the bread-and-butter business of chemical industry since its very emergence. There are thousands of chemical providers worldwide: large and small, locally operating and international, young and mature, application-oriented and compound-focused, technology specialists, pure manufacturers, traders, refiners, hybrids, and companies working on demand while others manufacture for the free market.
So, what is special about ChiroBlock's "SupplyChainFactory"?
In fact: Nearly everything, starting from the internal structuring and positioning. The "SupplyChainFactory" is ChiroBlock's youngest business branch originating from the other three R&D-oriented business fields and hence being a "downstream" activity that is always based on the results of our R&D. The overwhelming majority of other classical chemical companies regards manufacturing as their core business and only opportunistically add some R&D activities if regarded appropriate.
- We synthesize only new, otherwise commercially not available fine and special chemicals. Others produce known standard chemicals.
- We manufacture only customer-exclusively. Others additionally offer chemicals via catalogues or databases.
- We are focused on scales between some milligrams and a few kilograms. Others regard this scale as being too small / not profitable.
- We manufacture these small amounts under a strict QM-system (DIN EN ISO 9001, IPEC 2006PQG GMP). Others often spare the costs of a QM system for such small amounts of products.
- We welcome (customer) audits even for low-volume products. Others refuse (customer) audits for such products because of their costs.
- We manufacture at the same and only site where the entire R&D work had been done before - in the middle of Europe and Germany. Others often separate R&D from manufacturing locations with consequences for supply chain security, communication, and quality.
- We also synthesize sophisticated, complex, difficult to make compounds. Others are mainly focused on simpler compounds with feasible synthetic approaches or on a small number of synthetic operations/compound classes.
- We have the resources to (fundamentally) optimize a running process at any time. Read more about our ReDesignFactory. Others, in most cases, only have access to resources for "technological process improvements" and lack the ability to improve the synthetic approach fundamentally (route, applied synthetic R&D).
- We only manufacture as long as the customer really requires it, since our business model strategically does not depend on "manufacturing". Others are bound to charge the production facilities to capacity, if necessary, also by producing non-exclusive products for the free market.
- We are happy to transfer the manufacturing process to third parties on customer's request. Others keep process details protected even from customer's eyes.
- We do not manufacture own "final products" - there is no inherent conflict of interests with customers. Others also manufacture "final products" causing a potential conflict of interests with customers.
- We are fully aware of the problems encountered when sourcing fine chemicals in small to medium amounts from a CMO at constant quality and on a reliable basis:
- Large companies have neither the infrastructure nor internal processes for offering this kind of service.
- On the other side of the scale, pure "synthesis labs" (e.g., those at universities or public institutes) lack the required QM system, sound and independent quality control, as well as efficient processes. When limited to the lab scale, syntheses become inefficient, costly, and lengthy if parallel work is required to meet a target amount exceeding some dozens or even hundreds of grams.
The SupplyChainFactory of ChiroBlock exactly fills this gap, allowing our supply chain customers to rely on first-class chemistry competencies, state-of-the-art facilities, and an audited quality management system.
Apart from normal lab-scale reactors we also have a fully equipped "kilo lab" (60-L reaction volume) to offer and can thus perform nearly all reactions and purification operations on a mg to multi-kg scale.
The scope of our capabilities comprises low-temperature reactions (down to –85 °C), inert gas syntheses, (semi)continuous flow reactions, photochemistry, certain high-pressure reactions, and chromatografic separations on a multi-hundred-gram scale. Our QM-regulated production manuals also address such important issues as:
- dedicated equipment
- line clearance
- sourcing and control of starting materials and intermediates
- safety assessment and risk minimization
- critical parameterso change management
- quality control
Since all of our "manufacturing processes" result from our R&D-oriented business fields, we are able to provide well-grounded information for establishing and obeying target specifications.
ChiroBlock's Manufacturing Process for fine & specialty chemicals
The very "genesis" of our manufacturing processes guarantees our thorough knowledge of both the target molecule and its synthesis. This, in turn, reduces the quality risk in production. Even in cases where the customer provides a synthesis process, we never directly apply it to produce the compound in question to avoid both sides facing problems and substantial follow-up costs that can be avoided if our typical approach is followed: 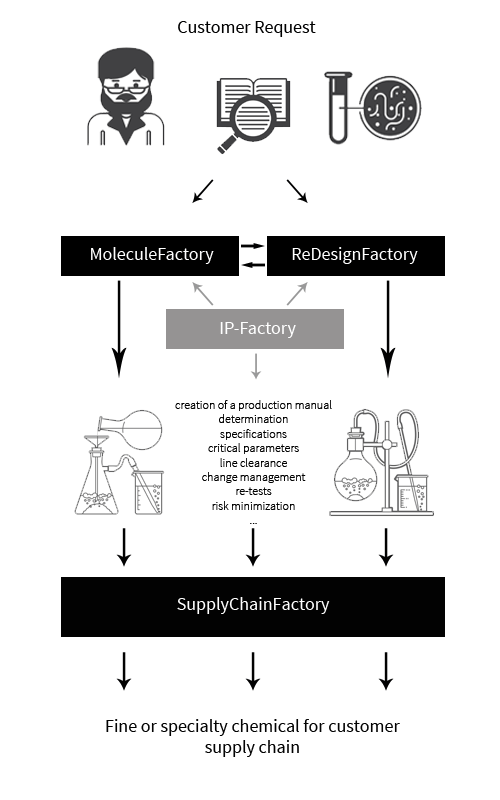
While this approach might seem complex at first sight, it was proven to be efficient, workable, robust, and overall advantageous over many years. Always bear in mind: We offer the SupplyChainFactory service as the missing link to industrial large-scale manufacturing and use it to prove the scalability and robustness of newly developed synthetic routes, thus utilizing this service as an answer to the sourcing needs for special, otherwise not available compounds.
However, this service cannot be regarded as being isolated from our other activities, since it is not just about selling resources and is not meant to fill our catalogue – we are ready to transfer any process to third parties, if requested.
There is no need for the SupplyChainFactory business to subsidize our R&D-focused activities as found elsewhere, since the latter are profitable on their own and - rather - the origin and basis of every repeatedly manufactured compound.
Some examples for our manufacturing service:
► A company of the consumer industry was looking for a new/second supplier for a special functional dye. Challenge: Both the target price and the target amount are limited by the first supplier. It was a tedious and expensive synthesis of a very crucial product for customer's supply chain.
We conducted a thorough synthesis optimization and managed a continious reliable delivery of the dye at lower internal costs and with improved quality (several kgs per year). After the first high-quality batches we offered to transfer the new process to the customer.
► A diagnostic company in need for a special polymer for an application in medical products. Challenge: High purity, narrow molecular weight distribution, sound analytical characterization required, a commercially available polymer of vague specification and an unknown manufacturing process (sold for applications in the construction industry). ChiroBlock established a robust synthesis procedure avoiding toxic reagents and a setup of an application-oriented target specification and analytical characterization. Today ChiroBlock manufactures several hundred grams per year for in vivo applications.
► An optoelectronic company in need of several highly pure products on scales of < 1 kg p.a. with very short product life cycles. They were searching for suppliers with focus on new and low-volume special chemicals. This search proved to be very difficult, especially in view of the additional requirements regarding the QM system and the "purity-by-design" approach. It was a close interaction of ChiroBlock's MoleculeFactory and SupplyChainFactory.
We developed a "purity-by-design" synthesis and purification strategies to match the specification requirements. ChiroBlock delivers reliably the target chemicals and has a short response time for the development of analogues.
show more examples of manufactured chemicals